|
The quality of the results will depend in large part on the quality of your printer. All of the tables will print on a single 8½ x 11 page, though some may require color to retain their appearance. The following descriptions include a sample element from each periodic table. The program can be downloaded from the Adobe Website. To view and print these files, you will need to install the free Adobe Acrobat Reader program on your computer. The simulation is produced by Columbia Public Schools, USA.Unless otherwise indicated, the following tables are in Acrobat PDF format. There is also some information on chemical bonds and a cool mystery elements challenge game. Interactive Periodic Table is an excellent way to explore and learn the Periodic Table. This means their outermost shell of electrons are fully occupied by electrons, which means there is no tendency for the atom to transfer or share electrons during chemical reactions, making it chemically stable. Noble gases have eight electrons in their outermost energy level, except for helium, which has only two electrons. They are unreactive because their atoms have stable arrangements of electrons. Group 0 (or Group 8) elements chemically unreactive The Periodic table starts at element atomic number 1 (this is Hydrogen, symbol H) and ends at element atomic number 118 (Ununoctium, symbol Uuo). Metals (like sodium and magnesium) appear on the left side of the Periodic Table, whereas Non-metals (like chlorine, helium and oxygen) appear on the right side of the Periodic Table.Īs you read along a Period (left to right), elements show increasing atomic number. Atoms of different elements have different numbers of protons.Įlements in the same group in the periodic table have the same number of electrons in their highest energy level (outer electrons) and this gives them similar chemical properties. 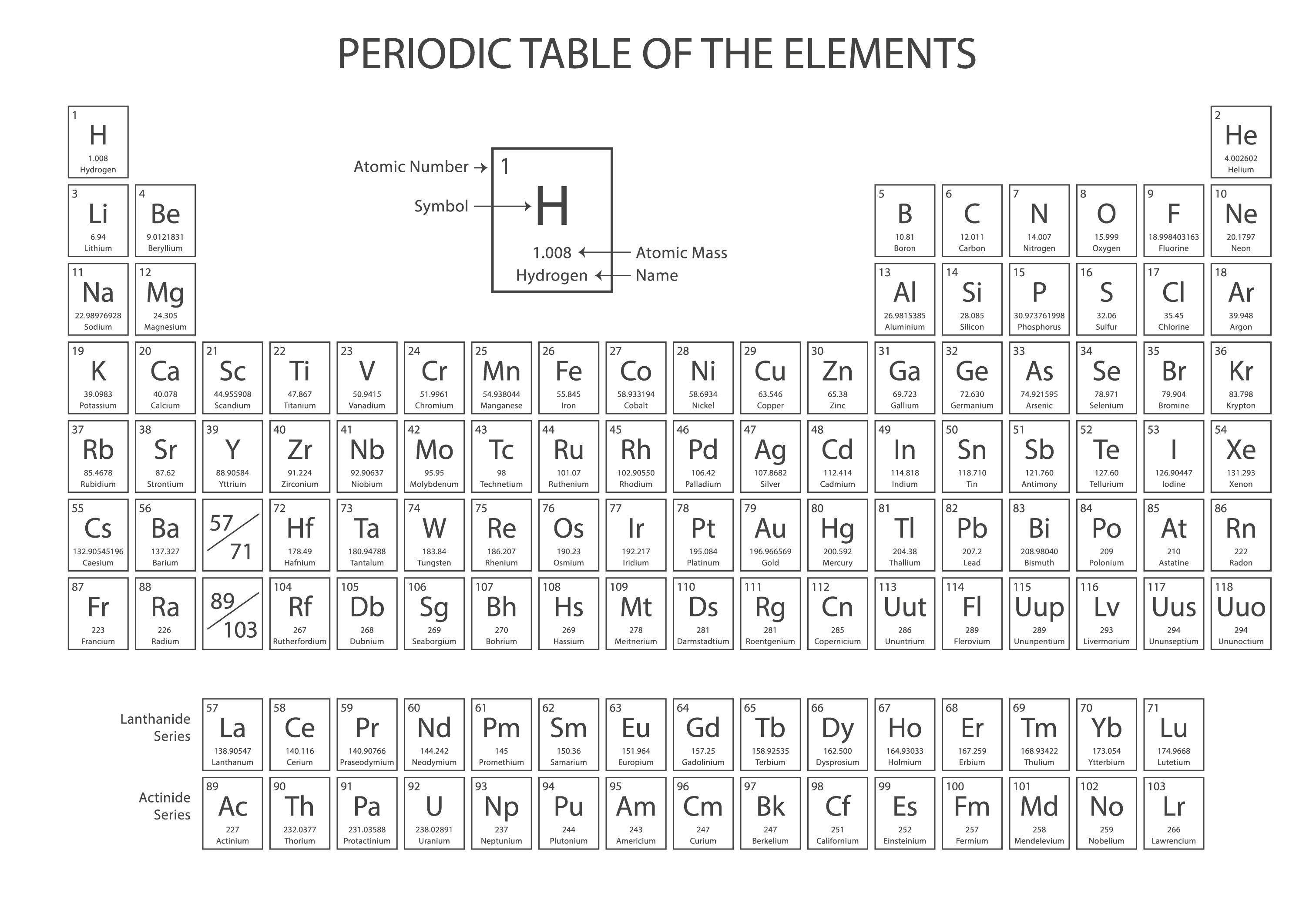
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
